Abstract
Background: Chimeric antigen receptor (CAR) T-cell therapy-associated hemophagocytic lymphohistiocytosis (carHLH) is an emerging toxicity/clinical syndrome associated with markedly elevated ferritin, coagulopathies, cytopenias, and multi-organ failure. The outcome of children who develop carHLH after CD19 CAR T cell therapy is poor. However, there is limited data on incidence and outcomes of adults with lymphoma who develop carHLH after CAR T therapy. Here, we report on the incidence and the outcomes of patient with LBCL who develop carHLH after axicabtagene ciloleucel (axi-cel).
Methods: We reviewed 164 consecutive patients with relapsed/refractory LBCL aged >/=18 years old who underwent standard of care axi-cel between December 27, 2017 and February 25, 2022 to determine the incidence of carHLH. CarHLH was definition by having 1 major and 2 minor clinical findings (as reported previously). The major criterion is ferritin greater than 10,000 or doubling within 1 week. Minor criteria are the development of grade 3 LFTs, LDH> >3 x ULN (U/L), hemophagocytosis in marrow, cytopenias needing transfusions, and fibrinogen <150 mg/dL.
Results: 7 patients (4.3%) met carHLH criteria. Patients who develop carHLH had a significantly elevated ferritin on Day 0 compared to those without (6,371.45 ng/mL vs 676.25 ng/mL p=0.005). Moreover, we found no significant differences for those with and without carHLH after analyzing gender, age, presence of bulky disease, LDH, those with or without double-hit, GCB vs. non-GCB, those with or without secondary CNS disease, and lines of therapy. All 7 carHLH patients experienced CRS at a median of 4 days [1-13], with 2 subjects having grade 1 and 5 subjects having grade 2; all 7 received dexamethasone and tocilizumab for CRS. 2 patients developed ICANS with max grade 1 and 4 at day 13 and 7 respectively.
In the 7 carHLH patients, the ferritin peaked a median of 8 days [2 - 25 days] after axi-cel infusion. As expected, those with carHLH patients had numerous laboratory abnormalities. The median and range are as follows: peak ferritin 11386 ng/ml [7556->100,000], peak AST 149 [84-2119], peak ALT 213 [31-1562], peak total bilirubin 2.1 [0.8-14.8], peak LDH 1562 [256- >1800], hemoglobin nadir 6.7 [4.3-8.3], and platelet nadir 5 [2-51]. We found that the average peak ferritin and peak LDH were higher in those who develop carHLH versus those that did not, 11386.5 ng/mL vs. 883 ng/mL, p<0.01 and 1562 U/L vs. 282 U/L, p<0.01, respectively.
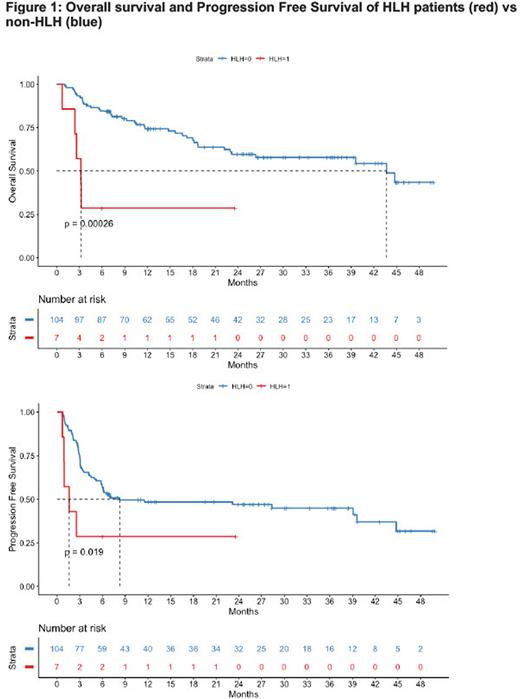
Notably, 4 out of the 7 carHLH patients had concomitant infections with presumed fungal pneumonia, covid pneumonia, CMV viremia, klebsiella pneumonia. Overall survival (OS) and progression free survival (PFS) and were significantly shorter in carHLH group with median OS of 3.2 months vs 44.3 months, p = 0.0026; and median PFS of 1.6 month vs 8.4 months, p=0.019, respectively after a median follow up of 17.7 months. Three patients with carHLH had progressive disease at day 27, 47, and 60. Overall, there were four deaths due to sepsis leading to multi-organ failure (n=1) and progressive disease (n=3).
Conclusion: This retrospective analysis demonstrates that carHLH is a rare (~4% in this study) but serious toxicity in a population of adult patients with LBCL who receive axi-cel. CarHLH was associated with the development of infections and inferior PFS and OS. Currently, there are no consensus treatment and prevent strategies for carHLH. Moreover, given the rarity of the syndrome, it is difficult to identify patients who are likely to develop carHLH prior to CAR T infusion. However, clinicians providing CAR T cell therapy should be aware of this emerging toxicity.
Disclosures
Arai:Kadmon: Membership on an entity's Board of Directors or advisory committees. Lowsky:Orca Bio: Research Funding. Meyer:GigaGen: Other: Co-founder, scientific advisor; Triursus Therapeutics: Other: Co-founder, scientific advisor; indee labs: Membership on an entity's Board of Directors or advisory committees; Orca Bio: Research Funding. Negrin:Magenta: Consultancy, Current equity holder in publicly-traded company; Garuda: Consultancy; Kuur: Consultancy; Amgen: Consultancy; UptoDate: Honoraria; Novartis: Consultancy; University of Pennsylvania: Other: DSMB or Advisory Board; CoImmune: Current equity holder in private company, Current holder of stock options in a privately-held company; BioEclipse Therapeutics: Current equity holder in private company, Current holder of stock options in a privately-held company. Shiraz:Kite, a Gilead company: Research Funding. Shizuru:Jasper Therapeutics: Consultancy, Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; rBio: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Shoreline BioSciences: Current equity holder in private company, Honoraria, Membership on an entity's Board of Directors or advisory committees. Sidana:Prothena: Honoraria; Magenta Therapeutics: Consultancy, Research Funding; Bristol Myers Squibb: Consultancy, Research Funding; Allogene: Research Funding; Janssen: Consultancy, Research Funding; Sanofi: Consultancy; Oncopeptides: Consultancy. Miklos:Fosun Kite: Consultancy, Honoraria; Kite, a Gilead Company: Research Funding; Janssen: Consultancy, Honoraria; Adaptive Biotech: Consultancy; Bristol Meyers Squibb: Consultancy; Novartis: Consultancy; Allogene: Research Funding; Pharmacyclics: Patents & Royalties: cGVHD Ibrutinib patent . Frank:Roche/Genentech - Wife: Current equity holder in private company, Current holder of stock options in a privately-held company; Kite/Gilead: Honoraria, Research Funding; Adaptive Biotechnologies: Consultancy, Honoraria, Research Funding; Allogene Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal